1Department of Radiodiagnosis and Interventional Radiology, All India Institute of Medical Sciences, New Delhi, India
2Department of Radiation Oncology, National Cancer Institute (NCI) campus, All India Institute of Medical Sciences, New Delhi, India
3Department of Pathology, All India Institute of Medical Sciences, New Delhi, India
Correspondence: Tej Pal
Received: 22 March, 2026; Accepted: 1 May, 2026; Published: 10 May, 2026
Citation: Saini M, Pal T, Meena SK, Mallick S, Hemlata (2026). Uncommon pathways: Spinal metastasis from intracranial anaplastic meningioma. Sci Academique 7(1), 49-56.
Abstract
Anaplastic meningioma is classified as a World Health Organization (WHO) Grade III tumor, notorious for its relentless progression, heightened mortality, and alarmingly frequent recurrences. Unlike typical meningiomas, which remain confined to the brain and meninges, anaplastic meningiomas break convention with their potential for extracranial invasion, though still rare. We present a case of 40-year-old male who presented with headache, limb weakness, and partial seizures. MRI revealed a right parasagittal mass, histologically confirmed as atypical meningioma. Despite surgical excision, recurrence necessitated revision surgery and radiotherapy. Five months post-treatment, the patient developed neck pain, and MRI revealed recurrent intracranial lesions and a large C2-C3 spinal mass causing vertebral destruction. Biopsies confirmed recurrent anaplastic meningioma with spinal metastasis. Although rare, extracranial metastasis from anaplastic meningioma is possible and can present significant clinical challenges. This case highlights the importance of maintaining a high index of suspicion for recurrence and distant spread in patients with high-grade meningiomas. Early detection and prompt management are crucial for improving outcomes in such aggressive and recurrent cases.
Introduction
Meningiomas are primary tumours that arise within the intracranial and intraspinal regions, and are typically benign in nature. They are often discovered incidentally, but when symptomatic, clinical presentation depends on the tumour’s size and location, manifesting as headache, nausea, vomiting, seizures, visual disturbances, hearing loss, or focal neurological deficits such as weakness. While the majority of meningiomas are benign, a small subset can behave aggressively and demonstrate malignant potential with the capacity to metastasize [1]. The types of meningiomas capable of metastasis include atypical (World Health Organization [WHO] Grade II) and anaplastic (WHO Grade III) variants [2], which together account for less than 5% of all meningiomas [3]. Specifically, anaplastic meningiomas represent approximately 2–3% of all cases and are characterized by rapid growth, high recurrence rates, and increased mortality [3]. Extracranial metastasis from anaplastic meningiomas is exceedingly rare, with an estimated incidence of only 1 to 5 cases per 1000 [4,5]. Among these, spinal metastasis has been documented in only a limited number of reports, highlighting the rarity of this clinical phenomenon [4,5].
Here, we present an unusual and noteworthy case of a patient initially diagnosed with atypical meningioma in 2023, who underwent complete surgical resection followed by adjuvant radiotherapy. During follow-up, the patient developed metastatic involvement of the vertebral column, illustrating the potential for aggressive progression even after standard treatment.
Case presentation
A 40-year-old male presented in August 2023 with complaints of headache, weakness in the left upper and lower limbs, and partial seizures. On clinical examination, he was found to be hypertensive, with mild weakness (Medical Research Council [MRC] scale 4) in the affected limbs. Magnetic resonance imaging (MRI) revealed a homogeneously enhancing dural-based mass measuring approximately 4 × 5 cm in the right parasagittal region. The patient underwent a right frontoparietal craniotomy with Simpson Grade II surgical excision. Intraoperative findings revealed a highly vascular, dural-based soft tissue mass infiltrating the adjacent brain parenchyma and densely adherent to the cortex. Histopathological analysis confirmed the diagnosis of atypical meningioma (WHO Grade II). Immunohistochemistry showed epithelial membrane antigen (EMA) positivity, focal CD99 positivity, and a Ki-67 proliferative index of >4 mitoses per 10 high-power fields (HPF).
During follow-up in January 2024, the patient again reported headaches. A repeat MRI revealed an ill-defined mass in the right parasagittal region measuring approximately 4.5 × 4 × 3 cm, accompanied by surrounding oedema (Figure 1-A), suggestive of tumour recurrence. In February 2024, the patient underwent a second right parasagittal craniotomy with gross total resection. Histopathological evaluation confirmed recurrent atypical meningioma, and the patient subsequently received adjuvant radiotherapy totalling 54 Gray across 27 fractions. Notably, during the course of radiation therapy, the patient’s limb weakness and pain improved significantly.
However, five months post-radiotherapy, the patient developed new symptoms, including persistent pain in the occipital and cervical region radiating downward. Despite receiving stepwise escalation of analgesics, his condition did not improve. An urgent MRI was performed, revealing two well-defined, T2 isointense extra-axial lesions—one in the right frontal lobe (approximately 1.3 × 1.4 cm) and another in the parietal lobe (approximately 2.8 × 2.2 cm)—both demonstrating homogeneous enhancement with adjacent dural involvement on post-contrast imaging (Figure 1 B, C, D, E). Additionally, a large lobulated, enhancing mass was identified cantered on the C2–C3 vertebral bodies (Figure 1 D, E & F), causing their destruction and extending anteriorly into the prevertebral space, posteriorly into paravertebral muscles, and into the spinal canal, resulting in significant spinal cord compression.
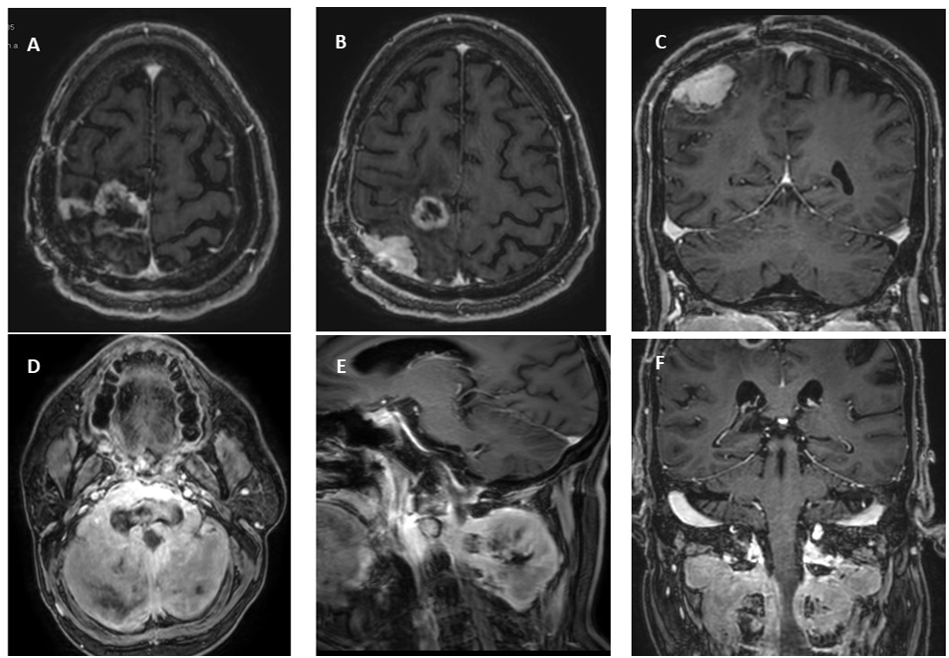
Figures 1: T1 Post Contrast sequences showing post treatment changes and recurrent lesion with spinal metastases. (A) Axial T1 MR sequence showing post treatment changes with peripheral nodular enhancement in right high parietal lobe in parafalcine location, (B) & (C) Axial and coronal sections showing recurrent meningioma along the dura in right parietal lobe (red arrows). (D), (E) and (F) Follow up scan in Axial, sagittal and coronal sections showing intensely enhancing soft tissue mass around C2- C3 vertebrae showing intraspinal extension and significant cervical spinal canal stenosis.
A provisional diagnosis of recurrent malignant meningioma with spinal metastasis was made. Biopsies were obtained from both the right frontal lesion and the cervical spinal mass. Histopathological evaluation of both specimens (Figure 2) confirmed anaplastic meningioma (WHO Grade III), characterized by marked nuclear atypia, high mitotic activity (26 mitoses per 10 HPF), and EMA positivity. Immunohistochemistry was negative for glial fibrillary acidic protein (GFAP) and S-100 proteins. These findings were consistent with high-grade anaplastic meningioma.
The patient was scheduled for gross total excision of the spinal lesion followed by radiotherapy. Unfortunately, he succumbed to the disease before the surgical procedure could be performed.
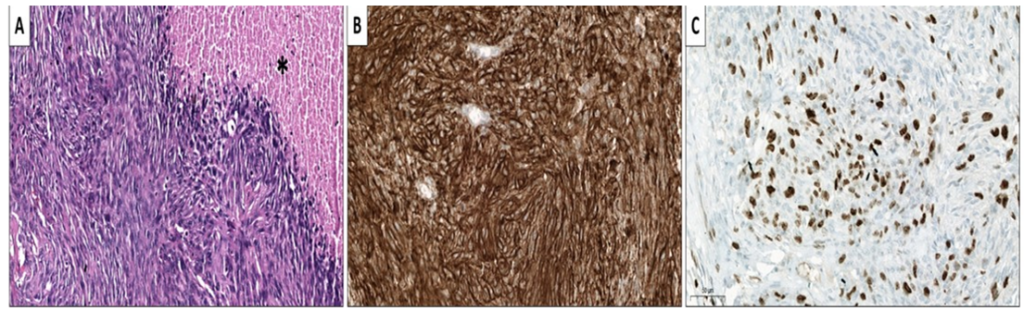
Figure 2: HPE images of C2- C3 mass biopsy specimen. Micrograph demonstrating (A) high grade histologic features of Anaplastic meningioma (WHO grade 3) with brisk mitotic activity, necrosis (Asterisks) and sarcomatous transformation (H&E 200x). (B) Tumor cells expressing Immunopositivity for EMA (IHC 400x) and (C) high MIB-1 labelling index (IHC 400x)
Discussion
Malignancy and extracranial metastasis in meningiomas are rare phenomena [4,5]. According to the WHO classification, meningiomas are histopathologically divided into three grades [6]. A higher propensity for extracranial spread has been documented in Grade II and III tumours [7,8]. Among the metastatic sites, the lungs are most frequently involved (60%), followed by the abdomen and liver (34%), cervical lymph nodes (18%), iliac, pelvic, and cranial bones (11%), pleura (9%), central nervous system (7%), and mediastinum (5%), Vertebral metastases are exceptionally rare, accounting for only 7% of reported cases [9]. To date, only a few cases of spinal metastasis from meningioma have been documented in the literature [10-12]. Immunohistochemical markers such as the Ki-67 proliferation index and CDKN2A (associated with 9p21 deletion) are valuable tools for assessing tumour aggressiveness, recurrence, and potential for metastasis [13].
Currently, there are no standardized treatment guidelines for metastatic meningioma [14]. Management typically includes a multimodal approach combining medical therapy, surgical resection, and radiation therapy, tailored to the individual patient’s condition and extent of disease.
Conclusion
This case highlights the rare but serious potential for anaplastic meningioma to metastasize extracranially, including to the spine. Early recognition of recurrence and distant spread is crucial for timely intervention. Regular follow-up with imaging is essential, even after complete surgical resection and adjuvant therapy. Immunohistochemical markers may aid in predicting aggressive behaviour. Given the lack of standardized guidelines, a multidisciplinary approach remains key in managing metastatic meningioma.
Disclosure
Funding: None
Date availability: All data and materials are available from the corresponding author, upon reasonable request.
Acknowledgements: None
Declarations
Conflict of interest: All authors declare that they have no competing interests.
Ethical approval: Ethical approval was not obtained for this as this is a case report.
References
- Modha, A., & Gutin, P. H. (2005). Diagnosis and treatment of atypical and anaplastic meningiomas: a review. Neurosurgery, 57(3), 538-550. https://doi.org/10.1227/01.neu.0000170980.47582.a5
- Apra, C., Peyre, M., & Kalamarides, M. (2018). Current treatment options for meningioma. Expert Review of Neurotherapeutics, 18(3), 241-249. https://doi.org/10.1080/14737175.2018.1429920
- Kessler, R. A., Garzon-Muvdi, T., Yang, W., Weingart, J., Olivi, A., Huang, J., Brem, H., & Lim, M. (2017). Metastatic atypical and anaplastic meningioma: a case series and review of the literature. World Neurosurgery, 101: 47-56, https://doi.org/10.1016/j.wneu.2017.01.070.
- Akimura, T., Orita, T., Hayashida, O., Nishizaki, T., & Fudaba, H. (1992). Malignant meningioma metastasizing through the cerebrospinal pathway. Acta Neurologica Scandinavica, 85(5), 368-371. https://doi.org/10.1111/j.1600-0404.1992.tb04063.x
- Conrad, M. D., Schonauer, C., Pelissou-Guyotat, I., Morel, C., Madarassy, G., & Deruty, R. (2001). Recurrent lumbosacral metastases from intracranial meningioma: report of a case and review of the literature. Acta Neurochirurgica, 143(9): 935-937, https://doi.org/10.1007/s007010170024
- Harter, P. N., Braun, Y., & Plate, K. H. (2017). Classification of meningiomas—advances and controversies. Chinese Clinical Oncology, 6 (Suppl 1). https://doi.org/10.21037/cco.2017.05.02
- Estanislau, E. S., Carvalho, G. T., Reis, B. L., de Freitas Barbosa, W., Brandão, R. A., Sousa, A. A., & Oliveira, J. B. (2009). Malignant meningioma with extracranial metastases. Arquivos de Neuro-Psiquiatria, 67(3A), https://doi.org/10.1590/s0004-282×2009000400036
- Thomas, R. Z., & Dalal, I. (2017). Extracranial metastases of anaplastic meningioma. BJR Case Reports, 3(2), https://doi.org/10.1259/bjrcr.20150092
- Fabi, A., Nuzzo, C., Vidiri, A., Ciccarese, M., Felici, A., Cattani, F., & Cognetti, F. (2006). Bone and lung metastases from intracranial meningioma. Anticancer Research, 26(5B), Google Scholar
- Palmer, J. D., Cook, P. L., & Ellison, D. W. (1994). Extracranial osseous metastases from intracranial meningioma. British Journal of Neurosurgery, 8(2), 215-228. https://doi.org/10.3109/02688699409027971
- Tominaga, T., Koshu, K., Narita, N., & Yoshimoto, T. (1994). Metastatic meningioma to the second cervical vertebral body: a case report. Neurosurgery, 34(3), 538-540. https://doi.org/10.1227/00006123-199403000-00024
- Tournat, H., Huchet, A., Ouhabrache, N., Thomas, I. C., Roubaud, G., & Maire, J. P. (2006). Bone metastatic evolution of a recurrent meningioma: case report. Cancer Radiothérapie, 10(8), 590-594. https://doi.org/10.1016/j.canrad.2006.06.008
- Terzi, A., Saglam, E. A., Barak, A., & Soylemezoglu, F. (2008). The significance of immunohistochemical expression of Ki-67, p53, p21, and p16 in meningiomas tissue arrays. Pathology Research and Practice, 204(5), 305-314. https://doi.org/10.1016/j.prp.2008.01.013
- Durand, A., Labrousse, F., Jouvet, A., et al. (2009). WHO grade II and III meningiomas: a study of prognostic factors. Journal of Neuro-Oncology, 95(3), 367-375. https://doi.org/10.1007/s11060-009-9934-0
