Ag and Zno: Antimicrobial and Cytotoxicity Activates Sarah Mohammad, Baghdadi Antonelli*, Fazia I Nolman
Mashhad Azad University, Iran
*Correspondence to: Baghdadi A
Citation: Mohammad S, Antonelli B, Nolman FI (2022) Ag and Zno: Antimicrobial and Cytotoxicity Activates. Sci Academique 2(2): 1-06
Received: 02 November, 2022; Accepted: 05 December 2022; Publication: 28 December 2022
Abstract
The leaves of Aloe vera were extracted by boiling in presence of stabilizing agent (Ammonia) to synthesize Ag nanoparticles at concentration 3.5mg/ml.The Ag NPs were recognized with UV- VIS spectrophotometer at optical density in (390, 400 and 450) nm respectively, also with a scanning electron microscope. The image was between 50-500 nm. The concentrations of nanoparticles measured using atomic absorption spectrophotometer which gave different concentrations of nanoparticles (3.5, 3.8 and 4) mg/ml of green synthetic of nanoparticles. AgNanoparticles solid and colloid Zn nanoparticles (Ag and ZnO) were used. Biosynthetic of these nanoparticles had antibacterial activity of Escherichia coli and Staphylococcus aureus with concentration of 0.17 mg/ml, while colloid nanoparticles was 3.5 µg/ml. The colloid ZnO nanoparticles showed antifungal activity on the tested fungi (Aspergillus niger, Pencillum spp and Candida albican) with concentration 0.17 mg/ml. The cytotoxicity effect of Aloe Vera boiling water extract and nanoparticles investigated. Results showed that the Aloe vera crude extract, biosynthetic Ag nanoparticles and ZnO showed significant effect on the HepG2 Cell line. Results showed that The Aloe vera extract at a concentration of 300 mg/ml and nanoparticles at a concentration of 3.5 mg/ml showed a healing effect in burns compared with silver sulfa- diagnosed as a control.
Keywords: Antimicrobial, cytotoxicity, nanoparticles, green Nanoparticles, Mice.
Introduction
The field of nanotechnology is one of the most active areas of research in modern material Nanotechnology is a field which developed day by day, making an impact in all. Sciences spheres of human life [1] and creating a growing sense of excitement in the life sciences especially biomedical devices and biotechnology [2]. Nanoparticles of noble metals such as gold, silver, platinum and zinc oxide are widely applied in products that directly come in contact with the human body, such as detergent, cosmetic products, and toothpaste, besides medical and pharmaceutical applications. Nanoparticle formation has been reported using chemical and physical methods. There are various methods for NPs synthesis such as sol-gel process, chemical precipitation, hydrothermal method, microwave and chemical and vapor deposition [3, 4].
Silver compounds have been used to treat burns, wounds infections [5].Various salts of silver and their derivatives are used as antimicrobial agents [6]. Previous studies have reported that Nano sized silver particles exhibit antimicrobial properties [7, 8] .Nanoparticles of silver have been studied as a medium for antibiotic delivery, and to synthesize composites for use as disinfecting filters and coating materials [9, 10]. Several mechanisms have been proposed to explain the inhibitory effect of silver nanoparticles on bacteria. The inhibitory activity of silver ions is higher in case of gram negative bacteria. This might be due to the thickness of the peptidoglycan layer in Gram-positive bacteria cell wall which may prevent to some extent, the action of the silver ions [11]. All these effects may impair the DNA replication which results in loss of cell viability and eventually resulting in cell death [12].The bactericidal effect of silver nanoparticles typically ranging from 2 to 5 nm has been investigated using green fluorescent protein (GFP)-expressing recombinant Escherichia coli [13]The silver nanoparticles of less than 10 nm diameters attached to the bacterial cell wall causes perforation of the cell wall, which leads to the cell death. There are some studies reporting that metal ions induce generation of intracellular reactive oxygen species in bacterial cells [14]. The highest sensitivity was observed against Methicillin resistant Staphylococcus aureus (MRSA) followed by Methicillin resistant Staphylococcus epidermidis (MRSE) and Streptococcus pyogenes. A moderate antimicrobial activity was observed in case of the gram negative pathogens Salmonella typhi and Klebsiellapneumonia([15]The colloidal silver particles, with variable sizes (44, 50, 35, and 25 nm), synthesized by the reduction of (Ag (NH3)2+ complexes with carbohydrates were tested for antimicrobial activity [16]. Therefore, the antibacterial activity was particle size dependent.
Materials and Methods
Media preparation: All media were prepared according to the instruction by manufactures supply.
Microbe isolates: All bacteria, fungi, and yeast isolates obtained from department of Biotechnology/Al-Nahrain University Iraq.
Animals used
Six groups of male mice with approximate weight of 20 g and aged 3-4 weeks used and distributed to eight mice in each group. These groups were:
- Positive Control: Wounded and treated with sliver sulfadiazine (SSD) at concentration of 1% silver sulfadiazine cream.
- Negative Control: Wounded but without any treatment.
- Group one: treated with grafted dressings content water extract of Aloe vera 300mg\ml
- Group two: treated with grafted dressings content green synthetic Ag nanoparticles 3.5mg/ml
- Group three: treated with grafted dressings content Ag colloid nanoparticles.3.5mg/ml ( Nanpar/ Iran) .
- Group four: treated with grafted dressings content ZNO colloid nanoparticles 3.5 mg /ml ( Nanpar/ Iran) .
Buffers for DNA extraction: All buffers and solutions for DNA extraction were prepared according to [18].
Cell Culture and Culture Conditions: HEPG2 cell line was used in this study were grown as a monolayer, in humidified atmosphere at 37°C with 5% CO2, spindle like cells. Cells were cultured in RPMI 1640 media supplemented with 10% FCS, 50 mg/ml streptomycin and 1000U/L penicillin. The experiments were performed when cells were healthy , at logarithmic phase of growth at passage 40.
Plant sample collection: Aerial parts of Aloe vera plants were cleaned under tap water. Then was freshly used for aqueous extract, and biosynthesis of silver nanoparticles [19]. The nanoparticles were identified by UV-visible spectrophotometer [20], for Scanning electron microscope[21] and for antimicrobial activity as in [22].
Results and Discussion
Results showed that the weight of the residue obtained after evaporation of water was 1.5 g, the appearance of the extract was brown color.
Biosynthesis of silver Nanoparticles: Results showed that the Silver nanoparticles were synthesized using the Aloe vera extract from silver nitrate (Figure 1)., which facilitates the formation of soluble silver complex Ag (NH3)2, which then facilitates the reduction. The reaction mixture turned pale yellow after 24 hrs. of reaction and exhibited an absorbance peak at 400 nm (Figure 2) which is a characteristic of silver nanoparticles due to its surface Plasmon absorbance. SEM analysis revealed that the silver nanoparticles were predominantly spherical (Figure 3), and the average size of the spherical silver nanoparticles was estimated to be 500nm. These results in agreement with [19] .In other study when they get the Ag [23]. A plenty of Ag+ were added into the extract of A. vera which contains many active small and large molecules. On one hand, the biological molecules might act as reducer to reduce Ag+ to be Ag NPs; On the other hand, they were also responsible for the stabilization of resulting nanoparticles

Figure 1: VIS absorption spectra of Ag nanoparticles in A. vera solution.
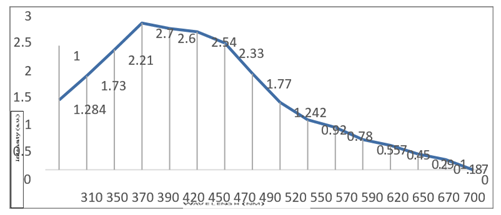
Figure 2: VIS absorption spectra of Ag nanoparticles in A. vera solution.

Figure 3: SEM sections of the green synthetic of Ag Nps Size (500).nm after 24hrs at 25C0 Characterization of Ag and ZnO colloid nanoparticles.
Ag nanoparticles were identified through the measurement of its wave length using UV\VIS spectrometer results in figures 2. The highest pecks were at 390 nm and 400 nm respectively Other properties (color, and size) of particles were used to identify the dark brawn solution with size (50-150) nm, these nanoparticles have high sensitivity to light which lead to change the orbital motion and result in changing the properties of Ag nanoparticles, whereas, ZnO nanoparticles were white solution with size (50-150) nm. These nanoparticles had the same sensitivity to light.
Antibiotic Sensitivity: Using The standard disk diffusion Results showed that E. coli was resistant to Ampicillin, Clindamycin and tetracycline, while it was sensitive to Chlaromphenicol, Norfloxacin, Nalidixic Acid, Cefotixime, Gentamicin, cephropive, Doxycycline and Azithromycin. While, S.aureus was resistant to Ampicillin, Azithromycin, Cephropive, Penicillin G, Clindamycin, Rifampin, vancomycin, Cephalothin, Nalidixic
| Antibiotic | Code | Concentration (μg/disk) | Sensitivity of E.coli | Sensitivity of Staphylococcus aureus |
| Ampicillin | AMP | 10 | R | R |
| iaehpmoralhC– col | C | 30 | S | S |
| nlhCiCeeCh G | G | 10 | R | R |
| Norfloxacin | NOR | 10 | S | S |
| Cephalothin | KF | 30 | R | R |
| Cefotixime | CTX | 10 | S | R |
| Clindamycin | DA | 30 | R | R |
| Gentamicin | CN | 15 | S | S |
| Tetracycline | TE | 10 | R | R |
| Vancomycin | VA | 30 | R | R |
| Azithromycin | AZM | 30 | S | R |
| cephropive | CE | 30 | S | R |
| Doxycycline | Do | 30 | S | S |
| Rifampin | RA | 30 | R | R |
| Nalidixic Acid | NA | 30 | S | R |
Table (1) Antibiotic Sensitivity test of E. coli and S. aureusto different antibiotics
Acid, cephotaxime and tetracycline, and it was sensitive to Gentamicin, Doxycycline, Norfloxcin and Chloromphenicol (table 1). In general it documented that very few of the conventional antibiotics are active against S. aureus. Many studies demonstrated multiple antibiotic resistance of S. aureus isolates, these isolates have multiple mechanisms for antibiotic resistance like inactivation of antibiotics by enzymes, modification of target site, impaired of penetration of drug target and present an efflux system [24]. From these results it was clear that E. coli was resistant to seven antibiotics, and sensitive to eight antibiotics. On the other hand S.aureus was resistant to eleven antibiotics and sensitive for four other antibiotics. Resistance to antibiotics may be caused by the antibiotic resistance genes; some of these genes may be located on chromosomal DNA, while others may be located on plasmid DNA as it was mentioned by [25]. Some reports indicated that S.aureus has continued to mutate and has developed intermediate resistance to Vancomycin which is acquired by mutation and thickening of cell wall due to accumulation of excess amount of peptyidoglycan .
Cytotoxicity effect of Aloe vera Extract and nanoparticles on Tumor cell line (HEPG2): The cytotoxic activity of A. vera water extract and nanoparticles determined by evaluating its effect on growth of HepG2 cell line after incubation for 72 hours at various concentrations of A. vera crude extract of 30, 15, 7.5,3.75, 1.8, 0.9 mg/ml) , Ag Nps (0.04, 0.02, 0.01, 0.005, 0,002 mg /ml), green synthetic Ag Nps and colloid ZnO (0.35, 0.175, 0. 87, 0.4, 0. 2, 0. 109, 0.05 mg \ml) Optical density of tumor cell line culture measured at transmitting wave length of 492 nm.
In the crude extract of Aloe Vera had a concentration independent effect on the cytotoxicity (figure 4, a). Cytotoxicity not increased with the increase of crude concentration. The highest effect of Crude extract was after 72 hrs. With certain concentrations, growth inhibition was increased significantly (P ≤ 0.05) effect at concentrations (0.93, 1.8 mg/ml) caused a significant growth inhibition to 57.33 %, 71% respectively. In figure (4 b) the results revealed a significant effect (p ≤ 0.05) at concentrations (0.05 and 0.109) of Ag Nps green, synthetic caused a significant growth inhibition to 57%, 71% respectively. In figure (4, c) showed that colloid ZnONps the present result revealed significant (p ≤ 0.05) at concentrations (0.2, 0.109 and 0.05), causing a significant growth inhibition to 42%, 44%, and 50% respectively Colloid Ag Nps showed in the result revealed significant (p ≤ 0.05) effect with concentration (0.43, 0.218, 0.109 and 0.0013 mg\ml), caused a significant growth inhibition to 40%, 70%, 67% and 48% respectively.
The anticancerogenic effects of the Aloe vera might be caused by the cytotoxic properties of these constituents, It was reported that A. vera extracts and Aloe emodin caused cytotoxicity and induced apoptosis in human cell lines CH27 (human lung squamous carcinoma cell), H460 (human non-small-cell lung carcinoma), HepG2 and Hep3B (2 human liver cancer cells), neuroectodermal tumor cells, and mouse lymphoma cells [26, 27] The hypothesis that ZnO NPs induce toxicity in a cell-specific manner was determined in the present study,ZnO NPs significantly altered the oxidant/antioxidant status of human liver cancer cells. [28]. Observed an acceleration of cell proliferation of HepG2 cells at very low concentrations of AgNP. The increase in cell number may be an initial effort by the liver to promote its detoxifying capacity after exposure to manageable levels of potentially toxic substances.
References
- Jobitha, G.G. and S. Rajeshkumar, Corresponding Author: G. Annadurai 3. J. Environ. Nanotechnol, 2013. 2(1): p. 20-27.
- Sharma, V.K., R.A. Yngard, and Y. Lin, Silver nanoparticles: green synthesis and their antimicrobial activities. Advances in colloid and interface science, 2009. 145(1): p. 83- 96.
- Pârvulescu, V.I., et al., Sol–gel-entrapped nano silver catalysts-correlation between active silver species and catalytic behavior. Journal of Catalysis, 2010. 272(1): p. 92-100.
- Dunn, K. and V. Edwards-Jones, The role of Acticoat™ with nanocrystalline silver in the management of burns. Burns, 2004. 30: p. S1-S9.
- Russell, A., et al., 7 Antimicrobial Activity and Action of. Progress in medicinal chemistry, 1994. 31: p. 351.
- Petica, A., et al., Colloidal silver solutions with antimicrobial properties. Materials Science and Engineering: B, 2008. 152(1): p. 22-27.
- Rai, M., A. Yadav, and A. Gade, Silver nanoparticles as a new generation of antimicrobials. Biotechnology advances, 2009. 27(1): p. 76-83.
- Ruparelia, J., et al. A comparative study on disinfection potential of nanosilver and nanonickel. Technical poster. in Proceedings of the 9th Annual Conference of the Indian Environmental Association (Envirovision-2006), entitled ‘‘Advances in Environmental Management and Technology”, Goa, India. 2006.
- Kim, S.H., et al., Low temperature synthesis and growth mechanism of Ag nanowires.
- Journal of Alloys and Compounds, 2007. 433(1): p. 261-264.
- Feng, Q., et al., A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. Journal of biomedical materials research, 2000. 52(4): p. 662-668.
- Gogoi, S.K., et al., Green Fluorescent Protein-Expressing Escherichia c oli as a Model System for Investigating the Antimicrobial Activities of Silver Nanoparticles. Langmuir, 2006. 22(22): p. 9322-9328.
- Ravishankar Rai, V. and A. Jamuna Bai, Nanoparticles and their potential application as antimicrobials. Science against microbial pathogens: Communicating current research and technological advances, A. Méndez-Vilas (Ed.), 2011: p. 197-209.
- Stohs, S. and D. Bagchi, Oxidative mechanisms in the toxicity of metal ions. Free Radical Biology and Medicine, 1995. 18(2): p. 321-336.
- Nanda, A. and M. Saravanan, Biosynthesis of silver nanoparticles from< i> Staphylococcus aureus</i> and its antimicrobial activity against MRSA and MRSE. Nanomedicine: Nanotechnology, Biology and Medicine, 2009. 5(4): p. 452-456.
- Panáček, A., et al., Silver colloid nanoparticles: synthesis, characterization, and their antibacterial activity. The Journal of Physical Chemistry B, 2006. 110(33): p. 16248- 16253.


